
Our Pipeline

Our Pipeline
We are targeting urgent unmet patient needs with our innovative approach.
ISAC
Gastric Cancer, Gastroesophageal Cancer
Phase 1
agonist
Non-small Cell Lung Cancer, Melanoma, & Other Solid Tumors
Phase 1
Colorectal, Non-small Cell Lung, Pancreatic, & Gastric Cancers
Preclinical
Solid Tumors Resistant to Checkpoint Inhibitors
Preclinical

Funds 3 bispecific Boltbody ISACs through early clinical development

(Caprin-1 ISAC)
Funds Boltbody ISAC targeting Caprin-1 through early clinical development
BDC-4182
BDC-4182 is a next-generation BoltbodyTM ISAC targeting claudin 18.2, a clinically validated target with expression in gastric cancer, gastroesophageal junction cancer, pancreatic cancer, and other tumor types. In preclinical models, BDC-4182 demonstrated significant anti-tumor activity in low claudin 18.2-expressing cancer models, induced immunological memory, and outperformed cytotoxic claudin 18.2 ADCs. Our next-gen Boltbody ISAC BDC-4182 is under development for gastric and gastroesophageal cancers. Enrollment is open for the Phase 1 dose-escalation trial. Learn more at Clinicaltrials.gov.
BDC-4182
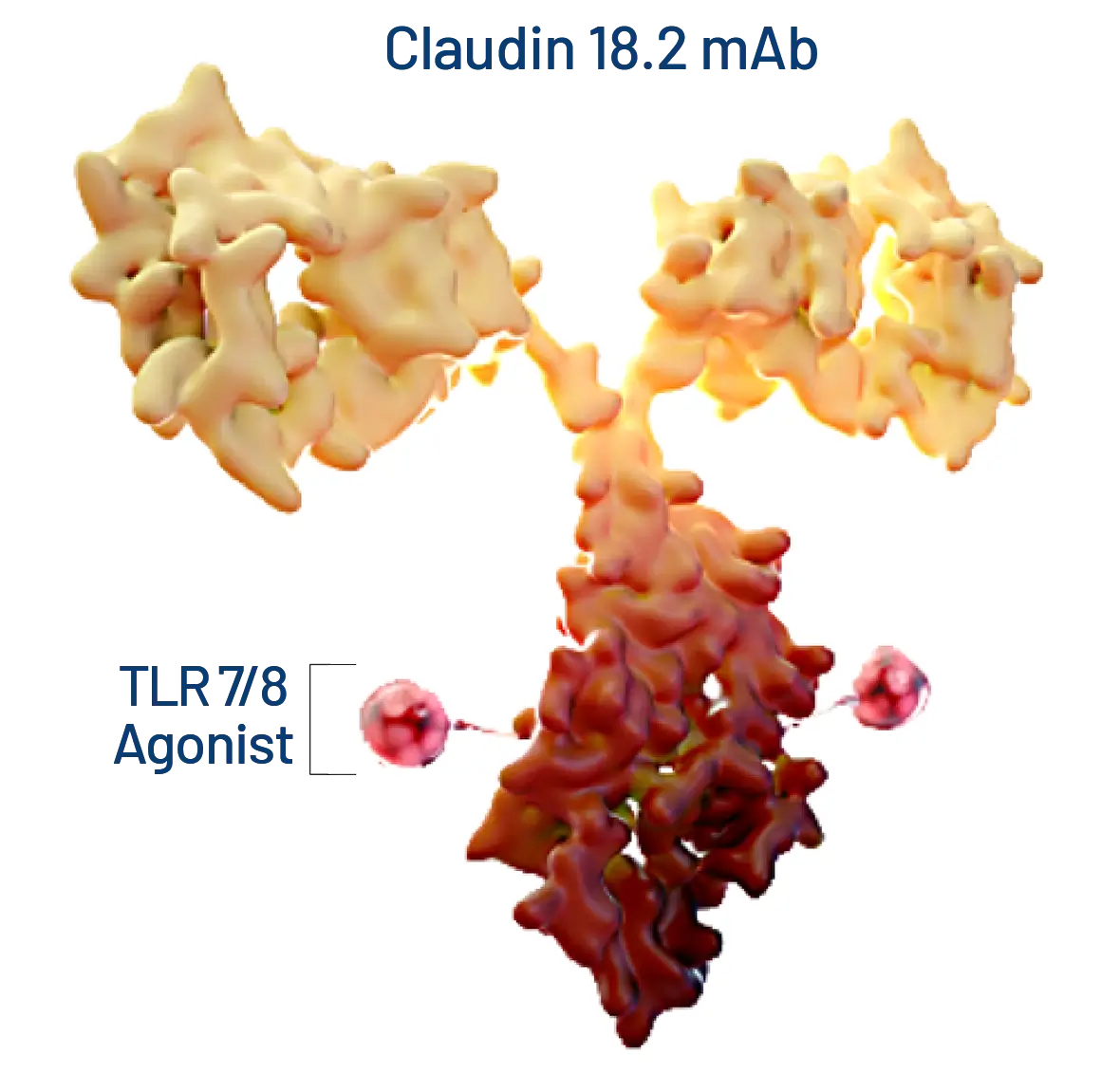
BDC-4182 Opportunity
- Clinically validated target in gastric cancer
- Large addressable market
- First-in-human clinical trial ongoing
Key Attributes
- Activity in low-antigen-density tumors brings hope to new patients
- Immunological memory protects against tumor re-challenge and recurrence
- Dramatically more potent than BDC-1001 in preclinical assays
Differentiation in Claudin 18.2 Landscape
- Superior efficacy versus TOPO1 and MMAE-ADCs in multiple experiments
- Potential for longer durable responses and recurrence prevention
- Safety benefit seen preclinically versus cytotoxic ADCs
